FFP3 Masks (10) White
Disposable Folded Respirator – FFP3 NR
Filtration Efficieny: BFE-PFE ≥ 99%
Anti-Fog
Prices exclusive of vat.
€19.85 Original price was: €19.85.€10.98Current price is: €10.98.
White Anti-Fog FFP3
Shipping 1-2 Business Days
Trading for over 25 Years
Standards:
- EN149 : 2001 + A1 : 2009 Respiratory protective devices – Filtering half masks to protect against particles – Requirements, testing, marking
- REGULATION (EU) 2016 / 425
- ISO 13485 : 2016 Medical devices – Quality management systems – Requirements for regulatory purposes
Model Number: MSK-22
Intended Use: These particulate respirators are designed to protect against solid and non-volatile liquid particles.
Fitting Instructions:
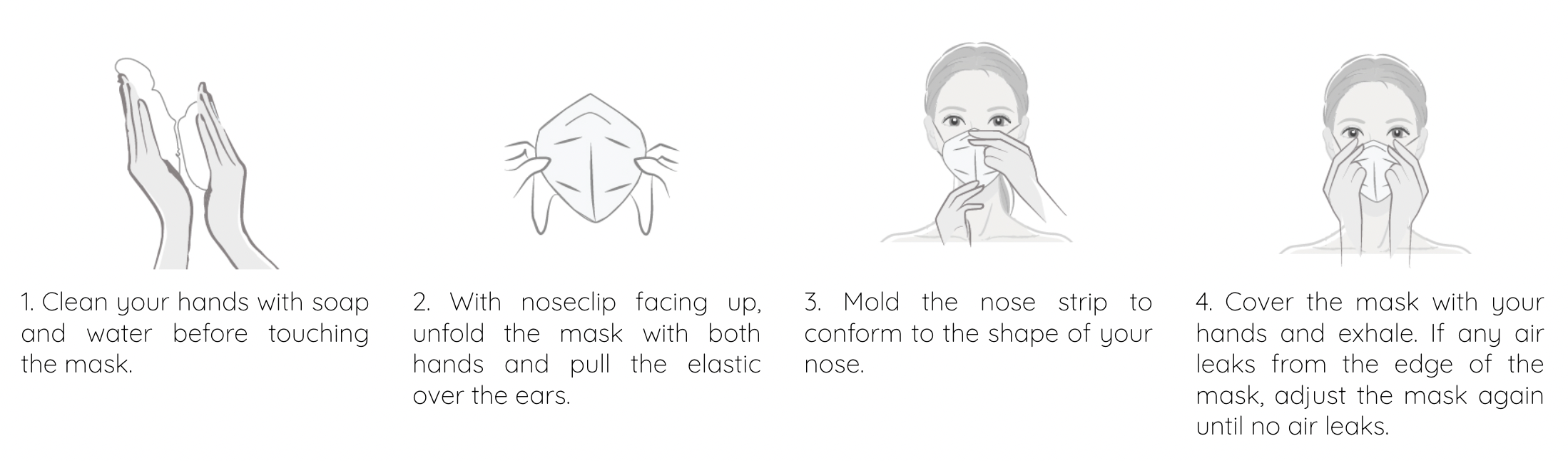
- 1. Hold the disposable folded respirator in position over the nose and mouth.
- 2. Pull the straps behind to ears, attach the straps to the retaining dip, improve comfort and prevent leakage
- 3. Ensure the nose clip is securely moulded around the nose, resting the ends against the cheek to obtain a good seal.
- 4. To check for proper fit, cup both hands over the mask and exhale vigorously. If air leaks around the nose, tighten the nose clip, if air leaks
- around the edge, reposition the straps for better fit.
- 5. Repeat adjustments until the mask is sealed properly.
- 6. If a proper seal cannot be achieved do not enter the contaiminated area or it may cause illness.
Storage:
1. Store in accordance with manufacturer’s instructions.
2. Storage temperature should be between -30 °C to +40 °C.
3. Storage humidity should be less than 80%.
4. When storing or transporting this respirator, use original packaging provided.
5. Do not store in direct sunlight.
Classification: There are three equipment classes : FFP1 /FFP2 /FFP3 ( see marking on box and on mask ). For the disposable folded Respirator marked “NR” representing Non-Reusable and shall be disposed after one shift.
Approvals: These respirators are tested by SGS institution, and audited by MNA LABORATORIES IND. TRADE. CO. LTD. These respirators are CE marked in accordance with the requirements of European Regulation (EU) 2016/425. The applicable legistraion can be determined by reviewing the certificate and Declaration of Conformity when requested.
Watch MUSK’s Promotional Video
Related Documents:
- Hibernia Medical does not supply goods on a sale or return basis.
- Hibernia Medical does not accept the return of goods without prior written communication to the Customer Service department.
- Goods can be returned once approval in writing has been given by Hibernia Medical.
- A ‘Goods Return Form’ must be completed for all returns. Notification of the intent to return Goods must be made within 14 days of the Delivery/Invoice date.
- The only goods that will accepted for refund must be a current stock line and not a discontinued line, nonreturnable product or a special-order item.
- All approved goods for return must be in perfect condition, in original packaging, sellable and be completely without defect or damage. Such damage will totally cancel any obligation by Hibernia Medical for any refund whatsoever on the item.
- Electrical and photographic equipment will only be accepted if complete with all leads, accessories and software. Any software must have its original seal intact.
- Goods are returned at the sender’s responsibility and cost. Hibernia Medical accepts no responsibility for goods lost or damaged during return transit. We recommend that the returned item(s) to our office are registered/tracked. Additionally, Hibernia Medical does not refund return carriage, postage fees or insurance costs.
- Please check the package and label before opening any products to ensure that you have received the correct items. Shipment errors must be reported no later than seven (7) days after the invoice date.
- Non-stock items and special-order items are subject to a no-return policy.
- All sales of clearance items are final and we cannot accept returns.
Please Note: Special order items and discontinued items are not subject to this returns policy and will be dealt with on a case-by-case basis.
All items returned are subject to a restocking fee of 10%.
Items returned after 14 days are liable to additional handling charges:
- 14 – 21 days old 15% of invoice value or minimum of €20.00
- 21 – 30 days old 20% of invoice value or minimum of €20.00
- 31 – 60 days old 25% of invoice value or minimum of €20.00
- Returns after 60 days are dealt with on a case-by-case basis
If you return part or all of your order, we will not refund the delivery charge as this part of our service to you has been completed. In the cases of incorrect, damaged or faulty goods, the delivery charge for a replacement to be sent out will not be charged.
For returns/refunds requests within 14 days please contact our Customer Service Team on 01 8665727 or email [email protected]
If you’ve asked for a refund, we will credit the account you used to pay for your order. Please allow up to 14 days for the refund to appear on your account.
Our Address:
Hibernia Medical
E3 Calmount Business Park
Ballymount
Dublin 12
D12 NA40
You May Also Like

Bosch

Littmann

Saver One

Omron

Omron

Iem

Braun

and

riester

mesi

medi Plent

sono site

wench allyn

plinth medical





