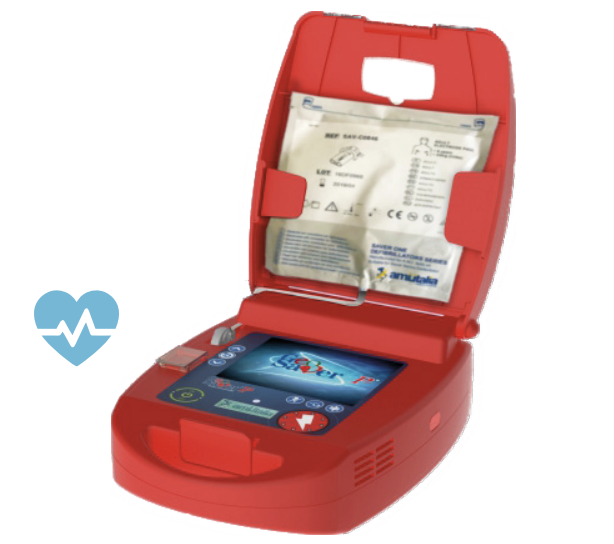
Geo Saver Fully-Automatic
Public access AEDs should be available everywhere, 24/7, in every city and community. The general approach to a prompt action in case of SCA should be spread to all communities; both institutions and citizens should be sensitized to play a decisive role for the first AID, while waiting for the arrival of EMS. Automatic external defibrillators should be clearly visible and recognizable in community centres, schools and public buildings/area to arise awareness on the importance of emergency preparedness.
€1,793.20 – €2,303.77
Shipping 1-2 Business Days
Trading for over 25 Years
GeoSaver is a portable Fully Automated External Defibrillator (AED) equipped with SIM /GPS/GPRS and a dual-battery powered system.
Practical and very simple to use. Advanced graphical interface combined with instructive voice prompt will guide the rescuers through each step for an effective lifesaving protocol. The VIVO BUTTON enable the rescuer to call EMS directly from the AED.
Everywhere to save lives GEO Saver AED’s are designed for a public access use and licensed to administer fast and safe rescues. Highly-effective and user-friendly for any lay rescuer, even without minimal training. The Fully automatic administers a defibrillating shock (when appropriate) with no shock button for the user to press whilst the Semi-Automatic administers a shock at the press of a button. Choose the best portable AED that’s right for you to save lives everywhere in any public circumstance (home, office, school, hotel, airport, train, beach, gym, pool, disco, etc.) and before EMS team arrives.
Automated testing to check daily functionality New design Several alternatives for recording and transfer data (internal memory, removable memory card, USB port, by remote) Equipped with two independent batteries: one dedicated to AED module and one dedicated to Geoloc module, to ensure that remote control feature does not drain the autonomy of the primary feature of defibrillator Biphasic technology up to 360J Unique feature: ECG can be sent in real time to AMISAVERCLOUD platform to enable another operator to monitor the ECG simultaneously with the local rescuer just trough any web connected device.
Automated testing to check daily functionality New design Several alternatives for recording and transfer data (internal memory, removable memory card, USB port, by remote) Equipped with two independent batteries: one dedicated to AED module and one dedicated to Geoloc module, to ensure that remote control feature does not drain the autonomy of the primary feature of defibrillator Biphasic technology up to 360J Unique feature: ECG can be sent in real time to AMISAVERCLOUD platform to enable another operator to monitor the ECG simultaneously with the local rescuer just trough any web connected device.
Meet AHA/ERC 2017 Guidelines Maintenance-Free: Automatically performs daily, monthly and six-month extensive self-checks of all main components: battery, internal electronics, energy charge and disarm, shock and ECG calibration systems. Daily testing data are stored by the device as text file (named AED1LOG) easily readable by any computer. AED runs further tests after each battery insertion as well as every time the device is turned on. A visual cue (green/red status indicator) provides effective alert to users whether AED is in working order and ready for a rescue.
- Service Mini-Screen: In standby the mini LCD screen displays a check mark confirming AED is ready for use and a battery gauge informing about the residual charge. Error codes will appear in faulty conditions.
- Helpful Menu: 3 buttons for navigating the software menu to set up device at user leisure: adjust the local date or time, adapt the screen or volume to ambient lights and noises, exclude the microphone while recording events, select a different language, print out the ECG files or simply get information on device and battery.
- CPR Coaching: More instructive voice and text prompts guide user through rescue. A built-in metronome assists responder during the CPR providing audio cues for the appropriate number and rate of chest compressions.
- Adult / Child Capability: Can be used on patients of any age with Adult or Paediatric proper electrodes. Device senses when paediatric pads are installed and automatically adjusts to use a more appropriate lower energy level (50J).
- Monitoring section menu: a new section has been introduced for the management of technical and physiological alarms and signals, according to IEC/EN 60601-2-27: patient loss, high or low heart rate, audio and visual signal for detection of a shockable rhythm so that the operator can switch/activate the semi-automatic modes to deliver the shock (using the appropriate pads); scaling of the ECG trace on the display (gain x2 or ÷2) reset of the audio or visual alarms.
A drone system that delivers Automated External Defibrillators (AEDs) to the scene of cardiac arrests. In doing so, bystanders will now have the ability to initiate life-saving measures while awaiting professional medical care. Out-of-hospital cardiac arrests affect some 275,000 individuals in Europe each year and carry a low survival rate of about 10%.
Research suggests when CPR and early defibrillation is initiated within the first few minutes the survival rate could potentially increase to as much as 70%.
See the video here: Everdrone Defibrillator Drone Video
Geo Saver Fully Automatic is the first geo defibrillator.
Designed for public access use and licensed to administer fast and safe rescues.
The shock, if required, will be delivered automatically whilst the VIVO BUTTON enable the rescuer to call EMS directly from the AED.
| Model Numbers: | |
| Code SGA-B0990: | Fully Automatic Standard Version at 200J |
| Code SGA-B0991: | Fully Automatic Power Version at 360J |
| Configuration Option/Box Content: | |
| Conf-Norm: Standard Basic Configuration | (adult pads, disposable battery) |
| Conf-Rech: Rechargeable Configuration | (adult pads, accumulator, charger station) |
Technical Data:
- Operation: Fully Automatic Version
- Energies: Standard max 200J or Power max 360J
- Waveform: Adaptive BTE (biphasic truncated exponential) conforming to patient chest’s impedance
- Protocols: Various adult shock protocols available on request
- Factory default: Adult Standard escalating 150, 200, 200J
- Adult Power escalating 200, 250, 360J
- Pediatric (Standard or Power) 50J fixed
- Charging time: ≤9 seconds with a new and fully charged battery depleted battery will result in a longer charging time
- Analysis time: IEC/EN 60601-2-4 from 4 to 15 seconds
- Impedance: 20-200 ohms
- Sensitivity: IEC/EN 60601-2-4 (AHADB, MITDB source), 97%
- Specificity: IEC/EN 60601-2-4 (AHADB, MITDB source), 99%
- Controls: 2 buttons: ON/OFF,
- “i“ info button
- Flashing Icons: “connects pads to patient“
- “adult/child“ informing on pads type use
- “don’t touch patient“ warning to stay clear
- “touch patient“ informing it’s safe to touch
- Indicators: Status LED indicator informing on device condition Battery gauge with remaining capacity rate
- Audible alerts and text display with service alarms
- Upgradeable: through a USB cable or memory card or by remote through AMISAVERCLOUD
Battery Option
Type: Li-SOCl2 Disposable, code SAV-C1032
Autonomy: 300 complete rescue cycles (shocks at 200J and CPR)
or 200 complete rescue cycles (shocks at 360J and CPR)
or 35 hours ECG analysis for a new and fully charged battery (*)
Shelf-Life: when stored in original packaging 5 years (*)
Battery-Life: 4 years once installed to AED, assuming one battery insertion test and daily self-test but without switching AED on (*)
Type: Li-ion Accumulator, code SAV-C1033
Recharging time: 2,5 hours with the charger station code SAV-C1034 (*) (recommended to charge every 4 months at least)
Autonomy: 250 shocks at 200J or 160 shocks at 360J or 21 hours in ECG analysis for a new fully charged accumulator (*)
Battery-Life: 2 years or 300 charging cycles (*)
Pads Options
Type: Disposable, pre-gelled and self-adhesive
Adult: Code SAV-C0846, for patient >8 years or >25 kg
Pediatric: Code SAV-C0016, for patient <8 years or <25 kg
Cable length: 120 cm
Shelf-Life: 30 months
Event Recording
Internal memory: up to 6 continuous hours of ECG and rescue events
Optional memory: Removable SD card; length of storage depends on card capacity: a 2GB card records up to 100 hours
Data recording: “AED1LOG“ text file with detailed self-test activity
“AEDFILES“ multimedia files with complete recorded information
Event review: “Saver View Express“ data manager software
Physical
Size: 29,5 x 23 x 11,5 cm
Weight +/- 2,65 Kg
Environmental
Operating temperature: 0°C to 55°C (32°F TO 131°F)
Storing/Shipping temperature: -40°C to 70°C (-40°F TO 158°F)
Humidity: 10% to 95% relative humidity non condensing
Sealing (IP Protection): IEC/EN 60529 class IP56; splash proof, dust protected
Shock/Drop Abuse Endurance: IEC/EN 60601-1 clause 21; 1 meter drop, impact, force, rough handling, mobile tolerance
Electrostatic Discharge: IEC/EN 61000-4-2
Electromagnetic Compatibility: IEC/EN 60601-1-2 Emission, Immunity
Electrical Protection: IEC/EN 60601-1; Internally Powered, Type BF
Directive 93/42/CEE and 2007/47/CE: Class IIb
Radio Equipment Directive (RED): Directive 2014/53/UE
Geoloc Module
Frequency: GSM: 850, 900, 1800, 1900 MHz;
UMTS: 900, 2100 MHz
GPS: 1575, 1600 MHz
Geoloc Battery Options
Type: Li-SOCl2 Disposable, code SAV-C1038
Shelf-Life: when stored in original packaging 5 years (*)
Battery-Life: 4 years once installed to AED, assuming one battery insertion test and daily self-test but without switching AED on (*)
Type: Li-ion Accumulator, code SAV-C1039
Recharging time: 2,5 hours with the charger station code SAV-C1040 (*)
(recommended to charge every 4 months at least)
Battery-Life: 2 years or 300 charging cycles (*)
(*)Temperature at 20°C Humidity 45% non-condensing
Related Documents:
- Hibernia Medical does not supply goods on a sale or return basis.
- Hibernia Medical does not accept the return of goods without prior written communication to the Customer Service department.
- Goods can be returned once approval in writing has been given by Hibernia Medical.
- A ‘Goods Return Form’ must be completed for all returns. Notification of the intent to return Goods must be made within 14 days of the Delivery/Invoice date.
- The only goods that will accepted for refund must be a current stock line and not a discontinued line, nonreturnable product or a special-order item.
- All approved goods for return must be in perfect condition, in original packaging, sellable and be completely without defect or damage. Such damage will totally cancel any obligation by Hibernia Medical for any refund whatsoever on the item.
- Electrical and photographic equipment will only be accepted if complete with all leads, accessories and software. Any software must have its original seal intact.
- Goods are returned at the sender’s responsibility and cost. Hibernia Medical accepts no responsibility for goods lost or damaged during return transit. We recommend that the returned item(s) to our office are registered/tracked. Additionally, Hibernia Medical does not refund return carriage, postage fees or insurance costs.
- Please check the package and label before opening any products to ensure that you have received the correct items. Shipment errors must be reported no later than seven (7) days after the invoice date.
- Non-stock items and special-order items are subject to a no-return policy.
- All sales of clearance items are final and we cannot accept returns.
Please Note: Special order items and discontinued items are not subject to this returns policy and will be dealt with on a case-by-case basis.
All items returned are subject to a restocking fee of 10%.
Items returned after 14 days are liable to additional handling charges:
- 14 – 21 days old 15% of invoice value or minimum of €20.00
- 21 – 30 days old 20% of invoice value or minimum of €20.00
- 31 – 60 days old 25% of invoice value or minimum of €20.00
- Returns after 60 days are dealt with on a case-by-case basis
If you return part or all of your order, we will not refund the delivery charge as this part of our service to you has been completed. In the cases of incorrect, damaged or faulty goods, the delivery charge for a replacement to be sent out will not be charged.
For returns/refunds requests within 14 days please contact our Customer Service Team on 01 8665727 or email [email protected]
If you’ve asked for a refund, we will credit the account you used to pay for your order. Please allow up to 14 days for the refund to appear on your account.
Our Address:
Hibernia Medical
E3 Calmount Business Park
Ballymount
Dublin 12
D12 NA40
You May Also Like
You may also like…
-
AED Defibrillators & Accessories
Savepads Electrodes| Selfadhesive| 1 Pair
€81.68ex VAT€81.68inc VAT

Bosch

Littmann

Saver One

Omron

Omron

Iem

Braun

and

riester

mesi

medi Plent

sono site

wench allyn

plinth medical